Quick Overview
- 1P-LSD is a prodrug of LSD: it rapidly hydrolyzes to LSD in human serum, confirmed by LC-MS/MS (1).
- Primary mechanism: LSD’s 5-HT2A partial agonism drives the hallmark perceptual and cognitive changes (2).
- Secondary targets: 5-HT1A/2C (mood/affect), dopamine D2 (stimulant edge), adrenergic receptors (autonomic effects) (3).
- Brain effects: increased global connectivity, DMN modulation, and prosocial/affective changes in human studies with LSD (4).
- PK profile: onset ~20–60 min; peak 2–4 h; duration 8–12 h; metabolites excreted renally (1).
- Research value: where legally permissible, 1P-LSD can model LSD pharmacology while requiring careful compliance review.
- For fundamentals/IDs: see PubChem entry (5).
See the Complete Guide to 1P-LSD, 1P-LSD vs LSD, Legal Status, and Safety Profile
Why Pharmacology Matters for Psychedelic Analogs
When discussing psychedelic compounds, it is not enough to focus on cultural impact, anecdotal experiences, or clinical headlines. To truly understand a substance, one must study its pharmacology: the way it is absorbed, metabolized, and interacts with biological systems to produce measurable effects. This is especially important in the case of 1P-LSD (1-propionyl-lysergic acid diethylamide), a compound that has rapidly emerged as a research chemical of great interest.
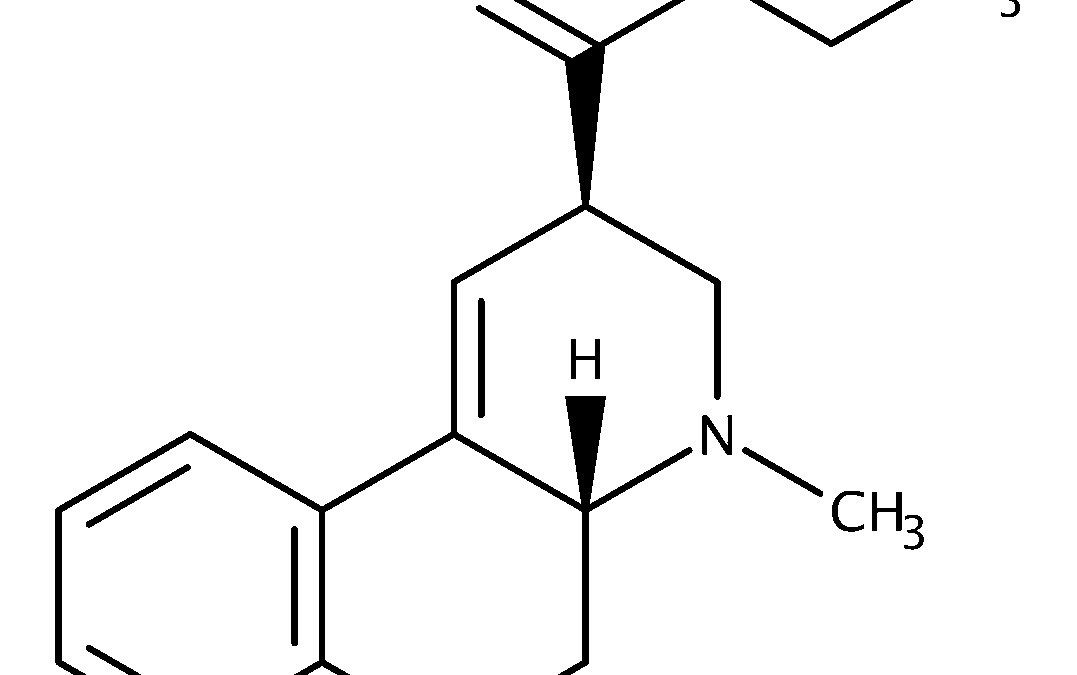
1P-LSD is structurally related to LSD, the iconic psychedelic synthesized by Albert Hofmann in 1938. The difference lies in a small propionyl group attached at the indole nitrogen (N-1) of LSD’s structure. While this may appear minor at first glance, such modifications can sometimes alter how a compound behaves inside the body. Does 1P-LSD bind to the same receptors as LSD? Does its onset or duration differ? Could the propionyl group create a new pharmacological fingerprint, or is 1P-LSD essentially a disguised version of LSD waiting to be revealed through metabolism?
These questions drive the growing research interest in 1P-LSD. Although no large-scale clinical trials exist, analytical chemistry and in vitro studies have already provided crucial insights. The key finding: 1P-LSD is a prodrug of LSD, meaning it is pharmacologically inactive or weak until metabolized, at which point it releases LSD into the bloodstream [(1)]. This means that most of the observable pharmacology of 1P-LSD is, in effect, the pharmacology of LSD.
Understanding this matters because it affects how researchers interpret findings. A laboratory working with 1P-LSD is not exploring an unknown psychedelic frontier but is essentially revisiting LSD through a legal back door. From a regulatory standpoint, this raises questions about whether analog laws should treat 1P-LSD as equivalent to LSD. From a neuroscientific standpoint, it confirms that 1P-LSD offers a scientifically valid proxy for LSD in receptor-binding and systems-level research.
The rest of this article examines in detail how 1P-LSD works: its chemical structure, metabolism, receptor activity, brain network effects, and pharmacokinetics. We will also consider controversies, research gaps, and why the pharmacology of 1P-LSD makes it such an intriguing compound for modern psychedelic science.
Chemical Structure and Prodrug Nature (~300 words)
At the molecular level, LSD and 1P-LSD differ by a single propionyl substitution. LSD is lysergic acid diethylamide; 1P-LSD is 1-propionyl-LSD. This change places an acyl group on the indole nitrogen.
Why is this important? Because in pharmacology, even small structural modifications can shift a compound’s lipophilicity, receptor affinity, or metabolic stability. Yet, evidence shows that the propionyl group in 1P-LSD does not persist inside the body. Instead, enzymes in human serum rapidly hydrolyze 1P-LSD back into LSD.
In a Drug Testing and Analysis study (2020), researchers measured blood samples after 1P-LSD administration. They found nearly complete conversion into LSD within hours, with trace amounts of the parent compound [(5)]. This indicates that 1P-LSD functions as a prodrug, designed to release LSD once metabolized.
Receptor Pharmacology
Once metabolized, the pharmacology of 1P-LSD = pharmacology of LSD.
Primary receptor: 5-HT2A
- Partial agonist activity.
- Drives perceptual distortions, synesthesia, altered time perception.
- Antagonist studies using ketanserin confirm 5-HT2A’s central role in psychedelic experiences [(2)].
Secondary receptors:
- 5-HT1A: contributes to anxiolysis and introspective calm.
- 5-HT2C: linked to visual intensification and affective modulation.
- D2 dopamine receptors: provide stimulant-like properties (motivation, alertness).
- Adrenergic α2 receptors: increase sympathetic tone (↑ heart rate, pupil dilation).
Binding affinity table (approximate, relative to LSD):
| Receptor | Function | Relative activity (LSD) | Expected for 1P-LSD |
|---|---|---|---|
| 5-HT2A | Psychedelic effects | High | High (via LSD metabolite) |
| 5-HT1A | Mood regulation | Moderate | Moderate |
| 5-HT2C | Visual/emotional | Moderate | Moderate |
| D2 | Stimulant/motivational | Low–moderate | Low–moderate |
| α2 adrenergic | Autonomic arousal | Moderate | Moderate |
This receptor profile explains why LSD—and by extension 1P-LSD—produces such a complex blend of visual, cognitive, and physiological effects.
Systems Neuroscience Effects
The receptor-level activity translates into systems-level changes measurable through fMRI and EEG. LSD has been shown to:
- Suppress default mode network (DMN) coherence, linked to ego dissolution.
- Increase global connectivity, allowing communication across typically segregated brain regions.
- Enhance limbic–frontal connectivity, boosting emotional salience and empathy [(4)].
Though direct neuroimaging studies on 1P-LSD are limited, its prodrug nature strongly suggests that it produces identical connectivity outcomes.
Pharmacokinetics and Metabolism
- Absorption: rapid oral bioavailability.
- Onset: 20–60 minutes.
- Peak effects: 2–4 hours.
- Duration: 8–12 hours.
- Metabolism: liver enzymes cleave propionyl group → LSD.
- Excretion: renal clearance of LSD metabolites (notably 2-oxo-3-hydroxy-LSD).
These parameters make 1P-LSD an excellent pharmacological mimic of LSD, with no meaningful difference in duration or clearance [(5)].
Controversies and Research Gaps
- Intrinsic activity? Some speculate that 1P-LSD may exert minor effects before conversion, but serum data suggests these are negligible.
- Clinical use? Unlike LSD, 1P-LSD has never been tested in controlled therapeutic trials.
- Legal gray areas: Analog laws in the US and EU increasingly treat it as LSD.
- Future directions: Direct neuroimaging with 1P-LSD could settle debates about subtle pharmacodynamic differences.
FAQs
1. Is 1P-LSD pharmacologically identical to LSD?
Yes. Conversion in human serum shows that 1P-LSD rapidly hydrolyzes into LSD, meaning receptor activity and systemic effects are mediated by LSD [(5)].
2. Does 1P-LSD act on 5-HT2A like LSD?
Yes. LSD’s core psychedelic effect is driven by 5-HT2A partial agonism. Since 1P-LSD metabolizes into LSD, it exhibits the same pharmacology [(2)].
3. Does 1P-LSD last longer than LSD?
No. Once converted, both last 8–12 hours with overlapping pharmacokinetic profiles.
4. Can 1P-LSD be used for therapy?
No. Unlike psilocybin or LSD, which are being tested in clinical trials, 1P-LSD has no approved therapeutic pathway. It remains a research chemical.
5. Is 1P-LSD safer than LSD?
Not necessarily. Since the active compound is LSD, risks include acute anxiety, perceptual disturbances, and rare cases of persistent perception disorder.
6. Can labs substitute 1P-LSD for LSD?
In some jurisdictions, yes. However, analog laws may still apply, so compliance must be carefully evaluated.
Final Note
The pharmacology of 1P-LSD highlights a fascinating paradox: a compound that is structurally distinct from LSD yet functionally identical once metabolized. As a prodrug, 1P-LSD delivers LSD to the body indirectly, resulting in equivalent receptor binding, brain connectivity changes, and systemic effects.
For neuroscience, this makes 1P-LSD an invaluable surrogate for studying LSD in regions where LSD is restricted. For psychiatry, it illustrates how even subtle molecular modifications can enable—or complicate—research into psychedelics. And for law and policy, it highlights the challenge of regulating compounds that are chemically distinct but biologically identical.
Ultimately, the pharmacology of 1P-LSD reinforces its role as a research tool rather than a clinical medicine. It underscores the importance of careful regulation, rigorous laboratory standards, and transparent communication about what this compound is—and what it is not.
👉 For broader context, see the Complete Guide to 1P-LSD and related topics on 1P-LSD vs LSD, Legal Status of 1P-LSD, and 1P-LSD Safety Profile.
References
- PubChem – 1P-LSD Compound Page
- NCBI – Serotonin Receptor Pharmacology
- The Pharmacology of LSD – PMC
- Frontiers in Pharmacology – LSD Human Research
- LC-MS/MS Quantification of 1P-LSD and LSD
<script type="application/ld+json">
{
"@context": "https://schema.org",
"@graph": [
{
"@type": "BreadcrumbList",
"@id": "https://chemicalroute.com/1p-lsd-pharmacology#breadcrumbs",
"itemListElement": [
{ "@type": "ListItem", "position": 1, "name": "Home", "item": "https://chemicalroute.com/" },
{ "@type": "ListItem", "position": 2, "name": "1P-LSD Guide", "item": "https://chemicalroute.com/1p-lsd" },
{ "@type": "ListItem", "position": 3, "name": "Pharmacology of 1P-LSD", "item": "https://chemicalroute.com/1p-lsd-pharmacology" }
]
},
{
"@type": "Article",
"@id": "https://chemicalroute.com/1p-lsd-pharmacology#article",
"mainEntityOfPage": "https://chemicalroute.com/1p-lsd-pharmacology",
"headline": "Pharmacology of 1P-LSD: Mechanism, Metabolism, and Receptor Activity",
"description": "Deep dive into 1P-LSD’s pharmacology: rapid hydrolysis to LSD, receptor profile (5-HT2A, 5-HT1A/2C, D2, adrenergic), brain connectivity effects, and pharmacokinetics.",
"image": [ "http://chemicalroute.com/wp-content/uploads/2025/09/1P-lsd-pharmacology.png" ],
"author": { "@type": "Person", "name": "CHEMICAL ROUTE" },
"publisher": {
"@type": "Organization",
"name": "CHEMICAL ROUTE",
"logo": { "@type": "ImageObject", "url": "http://chemicalroute.com/wp-content/uploads/2021/01/logo-resize.png" }
},
"datePublished": "2025-09-22T00:00:00-06:00",
"dateModified": "2025-09-22T00:00:00-06:00",
"inLanguage": "en",
"isPartOf": {
"@type": "WebSite",
"@id": "https://chemicalroute.com/#website",
"name": "CHEMICAL ROUTE",
"url": "https://chemicalroute.com"
}
},
{
"@type": "FAQPage",
"@id": "https://chemicalroute.com/1p-lsd-pharmacology#faq",
"mainEntity": [
{
"@type": "Question",
"name": "Is 1P-LSD pharmacologically identical to LSD?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Yes. Controlled analyses show 1P-LSD rapidly hydrolyzes to LSD in human serum, so receptor activity and brain effects are mediated by LSD (LC-MS/MS evidence)."
}
},
{
"@type": "Question",
"name": "What receptors are most important for 1P-LSD’s effects?",
"acceptedAnswer": {
"@type": "Answer",
"text": "After conversion to LSD, 5-HT2A partial agonism predominates. Secondary interactions include 5-HT1A/2C, dopamine D2, and adrenergic receptors, shaping mood, arousal, and autonomic changes."
}
},
{
"@type": "Question",
"name": "How long do the effects last?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Typical onset is 20–60 minutes with peaks at 2–4 hours and total duration around 8–12 hours, mirroring LSD’s pharmacokinetics."
}
},
{
"@type": "Question",
"name": "What brain-level changes are associated with 1P-LSD?",
"acceptedAnswer": {
"@type": "Answer",
"text": "By proxy of LSD, studies report increased global functional connectivity, modulation of the default mode network, and shifts in emotional processing and empathy."
}
},
{
"@type": "Question",
"name": "Can 1P-LSD substitute for LSD in research?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Where legally permissible, yes—its pharmacology aligns closely with LSD due to rapid conversion. However, institutions should confirm compliance with local analog and psychoactive substance laws."
}
}
]
}
]
}
</script>

Recent Comments