Brief Overview
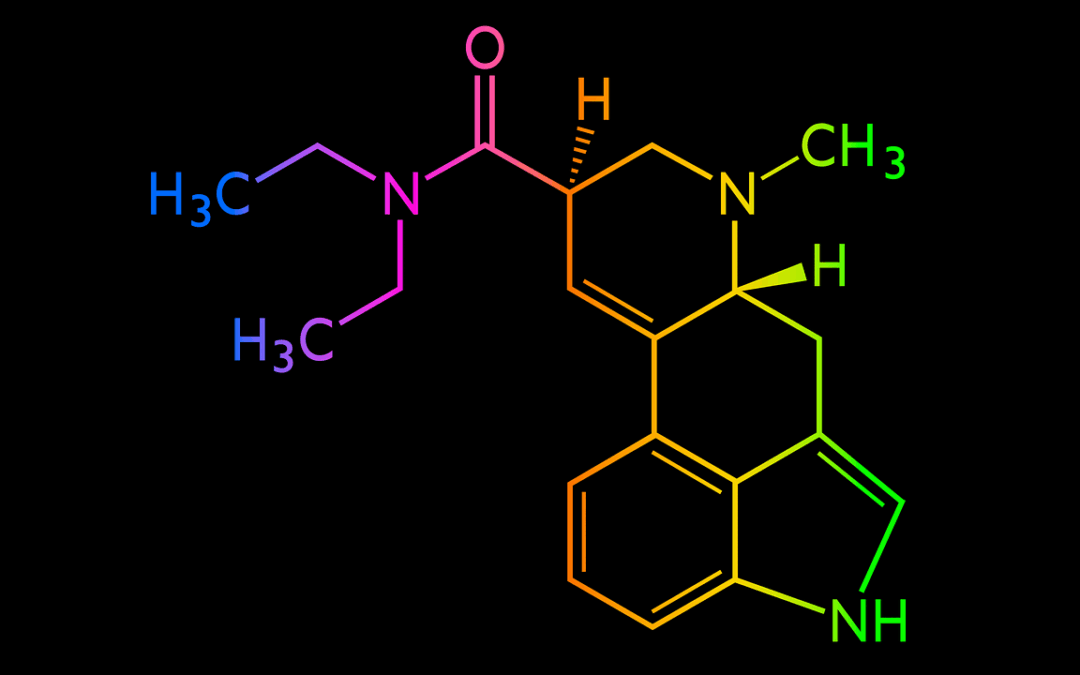
- Chemistry: 1P-LSD is an acylated lysergamide (propionyl group at N-1) that rapidly hydrolyzes to LSD in vivo, acting as a prodrug (1).
- Pharmacology: Both converge on LSD’s activity profile (notably 5-HT2A agonism) → similar perceptual and cognitive effects ([2], [6]).
- Effects: Onset and quality are broadly comparable; some users report slightly different timetables, likely due to prodrug conversion kinetics (research supports rapid conversion) ([1]).
- Legality: LSD is widely scheduled; 1P-LSD has faced tightening controls (e.g., UK PSA, Germany NpSG) or analog-law risk (US). See country snapshots below ([3], [7], [12]).
- Use in research: Where permitted, 1P-LSD offers a route to study LSD-like pharmacology; confirm local law before acquisition or study ([5]).
Introduction: Why Compare 1P-LSD and LSD?
1P-LSD and LSD sit next to each other on the lysergamide family tree. The practical question for researchers and policymakers is whether the N-propionyl modification in 1P-LSD meaningfully changes the science, safety, or legal posture compared with LSD. Evidence now indicates that 1P-LSD behaves as a prodrug—it’s quickly converted to LSD in serum and after administration—so many downstream effects overlap, yet compliance obligations and supply routes often differ by jurisdiction ([1]).
Chemistry & Biotransformation
Core structures
- LSD: diethylamide of lysergic acid (no acyl group at indole N-1).
- 1P-LSD: identical backbone with a propionyl group at N-1, classifying it as 1-acyl-LSD.
See structural registry and identifiers in PubChem ([1]).
Prodrug behavior of 1P-LSD
A controlled human/animal/serum study (Drug Testing and Analysis, 2020) demonstrated rapid hydrolysis of 1P-LSD to LSD after oral and IV administration, providing direct evidence that 1P-LSD is a prodrug of LSD (with high LSD levels and low residual 1P-LSD in serum). This mechanistic link explains similar onset/quality of effects and supports using 1P-LSD to model LSD pharmacology in settings where regulations differ ([1]).
Pharmacology: From Receptors to Networks
Receptor profile (convergent)
Once 1P-LSD is hydrolyzed, the active moiety is LSD, whose effects are mediated primarily via 5-HT2A receptor agonism; additional interactions include 5-HT1A/2C modulation, dopaminergic and adrenergic contributions. Reviews and receptor-level work consistently identify 5-HT2A as the principal driver of psychedelic phenomenology ([2], [6]).
Systems-level effects
LSD increases global functional connectivity and alters default-mode network dynamics; where studied, analogs that convert to LSD (like 1P-LSD) are expected to mirror these patterns given the shared active species. Human experimental work with LSD also maps prosocio-affective changes (e.g., empathy) with a strong 5-HT2A component ([6], [8]).
Subjective Effects & Time Course: Similar—With a Prodrug Twist
- Onset: Some users perceive a slightly slower onset for 1P-LSD; the DTA 2020 findings suggest rapid conversion nonetheless, so differences are often small and may relate to dose form, set/setting, or individual metabolism ([1]).
- Plateau & duration: Typically comparable for both, given the same active compound post-conversion.
- Qualitative profile: Visual enhancement, cognitive flexibility, time perception shifts, and emotional lability are broadly analogous when matched by dose and context ([2]).
Practical research note: For cross-compound comparisons, standardize dose units to estimated LSD equivalents after conversion to reduce confounds.
Safety & Risk Considerations (Research Context)
- Acute effects: anxiety, perceptual distortions, tachycardia, mydriasis—standard lysergamide profile.
- Persistent effects: rare HPPD and lingering anxiety reported for serotonergic hallucinogens; rigorous screening and harm-reduction protocols remain essential.
- Laboratory prudence: use certified standards, control environment, document chain of custody, and apply IRB/ethics oversight as required.
For context on hallucinogen safety frameworks and pharmacology, see peer-reviewed summaries ([2], [6]).
Legal Status: Where They Diverge Most
LSD
- Scheduled/prohibited in most jurisdictions (e.g., UN schedules; national narcotics laws).
1P-LSD
- United Kingdom: Controlled under the Psychoactive Substances Act 2016 (PSA)—production/supply/import are offenses regardless of specific scheduling ([3], [12]).
- Germany: Covered by the NpSG (New Psychoactive Substances Act) since 2019, restricting manufacture, trade, and supply of specified classes, including certain lysergamides ([5] general EU context; national NpSG applies).
- United States: Not explicitly scheduled as 1P-LSD federally; however, the Federal Analogue Act may apply if intended for human consumption—creating material enforcement risk ([7]).
- Broader EU/EU-DA monitoring: 1P-LSD and other NPS are tracked in the EU Early Warning System; annual reporting shows tightening controls and market shifts ([5]).
Research compliance tip: Treat 1P-LSD as high-risk from a legal perspective unless you have explicit, written clarity from local regulators/university counsel. Keep procurement strictly for research use and not for human consumption.
Practical Comparison: 1P-LSD vs LSD (Quick Table)
| Dimension | LSD | 1P-LSD |
|---|---|---|
| Chemical feature | Parent lysergamide | N-propionyl (1-acyl) lysergamide |
| Active species in vivo | LSD | LSD (via rapid hydrolysis; prodrug) [1] |
| Primary mechanism | 5-HT2A agonism (plus others) [2], [6] | Converges to LSD mechanism after hydrolysis [1] |
| Effects profile | Classic serotonergic psychedelic | Broadly similar at equivalent LSD exposure |
| Legal posture | Widely scheduled | Increasingly restricted (PSA UK; NpSG DE); analog-law risk (US) [3], [5], [7], [12] |
| Research use | Strict controls | Potentially accessible in limited contexts; verify local law |
(Citations are clickable in the references list.)
When to Choose 1P-LSD vs LSD (Research Decision Flow)
- Is LSD permitted under your institutional & national framework?
- Yes → Use LSD as the gold-standard reference compound.
- No → Proceed to step 2.
- Is 1P-LSD explicitly controlled in your jurisdiction?
- Yes → Do not procure without exemptions/permits.
- No/unclear → Seek written guidance from legal/compliance; ensure “research use, not for human consumption” procurement language and storage SOPs.
- Do you require equivalence to LSD’s receptor/network actions?
- Yes → 1P-LSD is acceptable only if you can justify prodrug conversion and standardize to LSD exposure using validated analytics (LC-MS/MS) ([13]).
- No → Consider alternative lysergamides with distinct profiles (e.g., ETH-LAD) if mechanistic differences are desired.
More Readings
- See our complete guide: 1P-LSD: Uses, Mechanism, Legality, and Safety
- You can also get more details:
- From Pharmacology → Pharmacology of 1P-LSD (Mechanism, Metabolism & Receptors)
- From Legality → 1P-LSD Legal Status: Country-by-Country Guide
- From Safety mentions → 1P-LSD Safety Profile: Risks, Side Effects & Harm Reduction
(Use descriptive anchors, not “click here”.)
FAQs
Is 1P-LSD basically the same as LSD?
Functionally similar after conversion—1P-LSD rapidly hydrolyzes to LSD in vivo, so the active species is LSD [(1)].
Does 1P-LSD have a slower onset?
Some users report a modest delay; the best evidence shows rapid conversion, so differences are typically minor and context-dependent [(1)].
Which is “safer”?
Neither is approved for human use. Risks reflect the serotonergic psychedelic class (acute anxiety, perceptual changes; rare persistent effects). Use rigorous lab controls and ethics oversight [(2), (6)].
Is 1P-LSD legal to buy for research?
Varies by country. Examples: prohibited under the UK PSA 2016, restricted under Germany’s NpSG, and potentially prosecutable under the US Analogue Act when intended for human consumption [(3), (12), (7)].
Conclusion
Chemically, 1P-LSD differs from LSD by a single propionyl group—but biologically and phenomenologically, both converge because 1P-LSD is a prodrug of LSD. For research teams, that means similar mechanisms and outcomes, with very different legal logistics. Validate your local framework, use certified analytics to verify conversion/exposure, and maintain robust lab governance.
References
- PubChem – 1-Propionyl-LSD compound page: https://pubchem.ncbi.nlm.nih.gov/compound/1-Propionyl-lysergic-acid-diethylamide; DTA 2020 (prodrug evidence): https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/10.1002/dta.2821
- Hallucinogens & 5-HT2A receptor (review): https://pmc.ncbi.nlm.nih.gov/articles/PMC5756147/
- UK Home Office PSA retailer guidance (Nov 2023): https://www.gov.uk/government/publications/psychoactive-substances-act-guidance-for-retailers/psychoactive-substances-act-2016-guidance-for-retailers
- Frontiers (LSD acute effects, affective findings): https://www.frontiersin.org/articles/10.3389/fphar.2021.711255/full
- EUDA/EMCDDA – European Drug Report 2025 (NPS overview): https://www.euda.europa.eu/publications/european-drug-report/2025/new-psychoactive-substances_en
- The Pharmacology of LSD (review): https://pmc.ncbi.nlm.nih.gov/articles/PMC6494066/
- CPS (UK) – Psychoactive Substances legal guidance: https://www.cps.gov.uk/legal-guidance/psychoactive-substances
- EUDA – European Drug Report 2025 (other drugs overview): https://www.euda.europa.eu/publications/european-drug-report/2025/other-drugs_en
- LC-MS/MS quant of 1P-LSD/LSD in biofluids: https://www.sciencedirect.com/science/article/abs/pii/S0731708519300135
- UK PSA 2016 (legislation.gov.uk): https://www.legislation.gov.uk/ukpga/2016/2/contents
<script type="application/ld+json">
{
"@context": "https://schema.org",
"@graph": [
{
"@type": "BreadcrumbList",
"@id": "https://chemicalroute.com/1p-lsd-vs-lsd#breadcrumbs",
"itemListElement": [
{ "@type": "ListItem", "position": 1, "name": "Home", "item": "https://chemicalroute.com/" },
{ "@type": "ListItem", "position": 2, "name": "1P-LSD Guide", "item": "https://chemicalroute.com/1p-lsd" },
{ "@type": "ListItem", "position": 3, "name": "1P-LSD vs LSD", "item": "https://chemicalroute.com/1p-lsd-vs-lsd" }
]
},
{
"@type": "Article",
"@id": "https://chemicalroute.com/1p-lsd-vs-lsd#article",
"mainEntityOfPage": "https://chemicalroute.com/1p-lsd-vs-lsd",
"isPartOf": { "@type": "WebPage", "@id": "https://chemicalroute.com/1p-lsd-vs-lsd" },
"headline": "1P-LSD vs LSD: Key Differences in Chemistry, Effects, and Legal Status",
"description": "Research-focused comparison of 1P-LSD and LSD covering chemistry, pharmacology, subjective effects, legality, safety and lab considerations.",
"image": [ "http://chemicalroute.com/wp-content/uploads/2025/09/LSD-VS-1P-LSD.png" ],
"author": { "@type": "Person", "name": "CHEMICAL ROUTE" },
"publisher": {
"@type": "Organization",
"name": "CHEMICAL_ROUTE",
"logo": { "@type": "ImageObject", "url": "http://chemicalroute.com/wp-content/uploads/2021/01/logo-resize.png" }
},
"datePublished": "2025-09-22T00:00:00-06:00",
"dateModified": "2025-09-22T00:00:00-06:00",
"inLanguage": "en",
"about": [
{ "@type": "Thing", "name": "1P-LSD" },
{ "@type": "Thing", "name": "LSD" },
{ "@type": "Thing", "name": "Lysergamide" },
{ "@type": "Thing", "name": "Serotonin 5-HT2A receptor" }
]
},
{
"@type": "FAQPage",
"@id": "https://chemicalroute.com/1p-lsd-vs-lsd#faq",
"mainEntity": [
{
"@type": "Question",
"name": "Is 1P-LSD basically the same as LSD?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Functionally similar after conversion: controlled studies show 1P-LSD rapidly hydrolyzes to LSD, so downstream pharmacology largely reflects LSD exposure. Research-use only; confirm local law."
}
},
{
"@type": "Question",
"name": "Does 1P-LSD have a slower onset?",
"acceptedAnswer": {
"@type": "Answer",
"text": "Some users report a modest delay, but experimental evidence indicates rapid conversion to LSD. Differences are usually small and context-dependent."
}
},
{
"@type": "Question",
"name": "Is 1P-LSD legal to buy for research?",
"acceptedAnswer": {
"@type": "Answer",
"text": "It varies by country. For example, the UK restricts supply under the Psychoactive Substances Act and Germany under the NpSG, while the US may prosecute under the Federal Analogue Act where intended for human consumption."
}
}
]
}
]
}
</script>

Recent Comments